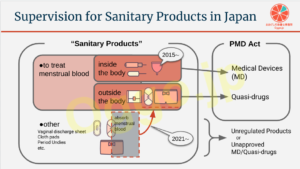
医療機器に「一般的名称:骨盤底筋訓練器具」が加わりました
「骨盤底筋を鍛える」という言葉を聞いたことがあるでしょうか。
この骨盤底筋は、緩んでいると腹圧性尿失禁が起こったりするとのことで(参考1)、改善のための骨盤底筋のトレーニングが勧められたりしています(参考2)。
女性向けの筋トレYoutubeにはよく登場しますし、整形外科の診療メニューにあったり、整体やリフレクソロジーでのサービスがあったりします。
骨盤底筋のトレーニング方には、身一つで行うやり方(ドローイング等)と、道具を用いるやり方(お腹あたりにEMSパッド貼ったり、膣内に挿入するいわゆる「膣トレグッズ」を使ったり)とが存在するようです。
医療機器としての骨盤底筋訓練器具を位置づける通知
実は先日2022年4月、骨盤底筋トレーニング業界(特に道具を用いる方)を揺るがす通知が2つ出されました。
URLを貼り付けておきます。
- ひとつめ:「題名長いので略(ざっくりいうと、厚生労働大臣の指定する医療機器の改正通知)」令和4年4月13日付、薬生発0413第1号、厚生労働省医薬・生活衛生局長通知
- ふたつめ:「骨盤底筋の運動に用いる機械器具の取扱いについて」(令和4年4月13日付、薬生監麻発0413第5号、厚生労働省医薬・生活衛生局 監視指導・麻薬対策課長通知)
この2つの通知により、骨盤底筋の運動に用いる機械器具は、下図のように整理されました。
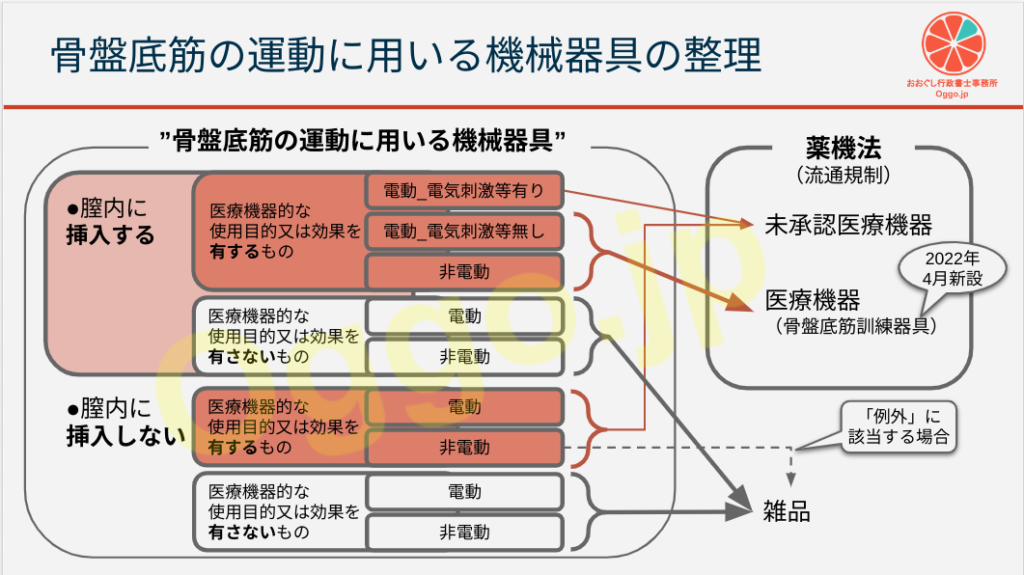
以下、ざっくりと通知の内容に触れていきます。
ひとつめの通知
ひとつめの通知では、一般医療機器(クラスⅠ)として以下の定義の「骨盤底筋訓練装置」が追加されたことが示されています。
家庭において、膣内に挿入して、尿漏れの改善等を目的とした骨盤底筋の訓練を行うための装置である。ただし、低周波や電気刺激等のエネルギーを与えるものを除く。圧力センサーや訓練内容を表示するプログラムを備えるものもある。
令和4年4月13日付、薬生発0413第1号、厚生労働省医薬・生活衛生局長通知
ふたつめの通知
ふたつめの通知では、骨盤底筋の運動に用いる機械器具の医療機器該当性について指針を示しています。
先程のパワポ画像に書いてある「医療機器的な使用目的又は効果」とは何なのかも、本通知にて「疾病の診断、治療若しくは予防又は身体の構造若しくは機能に影響を及ぼすこと」と示されています。
また続いて、医療機器とみなされる広告文言を例示で示してくれています。
次のような医療機器的な使用目的又は効果を広告・標ぼうするなど、医療機器的な使用目的又は効果を有するものは、医療機器に該当する。(中略)
【医療機器的な使用目的又は効果に該当する広告・標ぼうの一例】
「骨盤底筋の運動に用いる機械器具の取扱いについて」(令和4年4月13日付、薬生監麻発0413第5号、厚生労働省医薬・生活衛生局 監視指導・麻薬対策課長通知)
・ 損傷した骨盤底筋の修復
・ 尿漏れ(尿失禁)の治療、改善、予防
・ 子宮下垂、子宮脱の治療、改善、予防
・ マッサージ効果(疲労回復、血行をよくする、筋肉の疲れをとる、筋肉のこりをほぐすなど)
・ 鎮痛・消炎
・ 生理痛の緩和
・ PMS(月経前症候群)の改善
・ 更年期障害の緩和
・ 便秘の改善
・ 不眠症の改善
・ 冷え性の改善
・ 膣内の保湿
・ 膣分泌液の分泌機能改善
事業者さんへの影響
上述の例示には、骨盤底筋トレーニング器具として広告したくなっちゃう文言がずらりと並んでいるのが見て取れます。
でも、上記を広告する骨盤底筋トレーニング器具は、体内(膣)に挿入するしないに関わらず、医療機器とみなされるわけです。
医療機器であるならば、販売会社さんは販売前手続きを踏み、医療機器として流通させなくてはなりません。
それを怠った製品は未承認医療機器であり、それを流通させる行為は薬機法違反で逮捕もありえます。
事業者さんとしてはなかなか頭の痛い問題かと思いますが、ここはしっかり線引を理解して舵取りをしていかなくてはならないところです。
効果があるのならなおのこと、きちんとした形で消費者に届けなくてはいけません。
おわりに
弊事務所はフェムテックの分野でビジネスしていこう、と考える事業者の皆さまを応援しています。
医療機器にかかる一連の手続きについてはもちろん、医療機器にするしないの判断がつけられない!といったご相談もお受けしております。
最初の面談は無料ですので、企画段階からでも、手元に製品出来上がっていても、どうぞお気軽にご連絡ください。
参考
- "尿が漏れる・尿失禁がある". 日本泌尿器科学会. 更新日付不明. https://www.urol.or.jp/public/symptom/04.html. (参照2022.06.13)
- "骨盤底筋訓練". 東京女子医科大学附属足立医療センター 骨盤底機能再建診療部. 更新日付不明. https://twmu-amc.jp/mce/prsurgery/contents/training.html. (参照2022.06.13)
お気軽にご相談ください。
- 初回相談は無料です。
- 行政書士には秘密保持の義務が課せられております。
- フォームに入力されたメールアドレス以外に、当事務所から連絡差し上げることはいたしません。
“医療機器に「一般的名称:骨盤底筋訓練器具」が加わりました” に対して1件のコメントがあります。
コメントは受け付けていません。