家庭用検査キットの概要
オンライン診療が新型コロナウイルスの流行を契機に広がりを見せています。
そう遠くない未来には、「病気のときには病院に『行く』」という行為が変わってくるんだろうなと思います。
具体的には、病気のときにはまずオンラインで受診し、必要なときにのみ病院に出向くようになると予想しています。
さて、その変容のキーとなるものの一つに、「家庭用検査キット」があります。
文字通り、家庭で検査を行うためのキットです。新型コロナウイルスの検査キットでも注目を集めていますよね。
今後様々なものが出てくると想像していますが、とりあえず今回はその概要を簡単に書いてみようと思います。
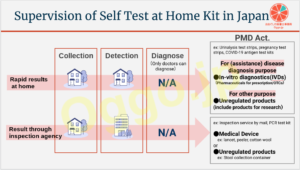
家庭用検査キットの薬事法上の整理
ここではまず、家庭用検査キットを、家庭で完結するものと、検査機関に検出を依頼するものとに分けてみます。
(いずれにおいても「診断」は行われません。これは医師にしかできないことだからです。)
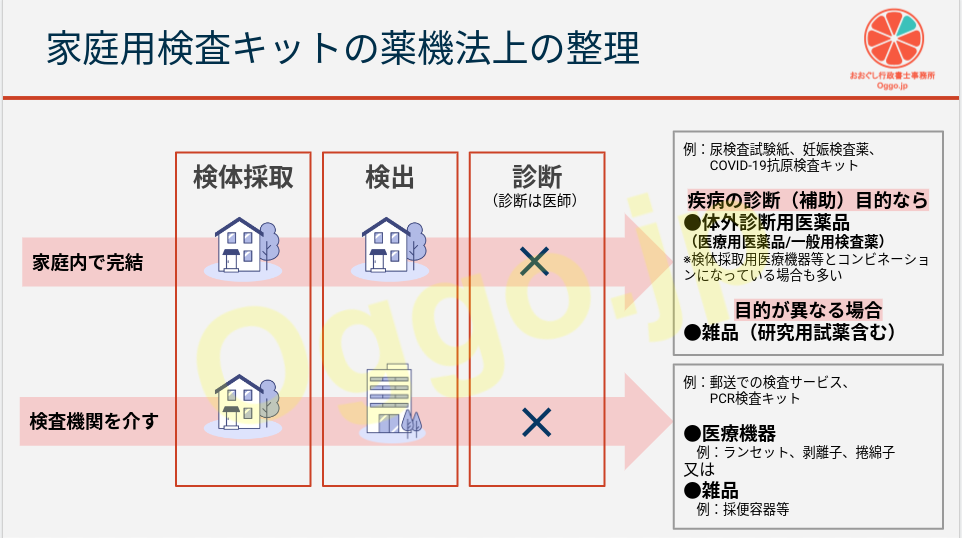
家庭内で完結する検査キット
短期間で検出結果がわかるものです。
例えば尿検査試験紙や妊娠検査薬そして新型コロナウイルスの抗原検査キットがこれにあたります。
使用目的により、薬機法上の取り扱いが変わります。
ざっくりいうと、こうです[1]。
![疾病の診断(を補助すること)が目的である→体外診断用医薬品
うち、医療用医薬品は、原則お医者さんに処方されないと入手できない。(COVID-19抗原検査キットを個人が薬剤師のいる薬局で購入できる[2]、一般事業者が医薬品卸売業者から購入できる[3]のは特例措置。)
一般用検査薬は現在4種類承認がある。[4][5]
尿糖、尿蛋白及び妊娠検査薬→第二類医薬品に分類→薬剤師または登録販売者が販売対応
尿中黄体形成ホルモン(LH)→第一類医薬品に分類→薬剤師が販売対応
疾病の診断(を補助すること)が目的ではない→雑品(研究用試薬を含む)](https://oggo.jp/wp-content/uploads/2022/08/image.png)
- 疾病の診断(を補助すること)が目的である→体外診断用医薬品
- うち、医療用医薬品は、原則お医者さんに処方されないと入手できない。(COVID-19抗原検査キットを個人が薬剤師のいる薬局で購入できる[2]、一般事業者が医薬品卸売業者から購入できる[3]のは特例措置。)
- 一般用検査薬は現在4種類承認がある。[4][5]
- 尿糖、尿蛋白及び妊娠検査薬→第二類医薬品に分類→薬剤師または登録販売者が販売対応
- 尿中黄体形成ホルモン(LH)→第一類医薬品に分類→薬剤師が販売対応
- 疾病の診断(を補助すること)が目的ではない→雑品(研究用試薬を含む)
体外診断用医薬品に該当する場合は、薬機法に基づく許可及び承認手続きが必要となります。手間はかかりますが審査により品質は確保されますし、また保険適用の可能性もあります。
研究用試薬とはなんぞや、となる方もいるかも知れませんが、これは要は理科室で使ってたリトマス試験紙と同じ、と思っていただくとわかりやすいかと思います。
品質は各メーカー頼りとなります。
ここあたりの品質の担保、つまり結果の信用度の都合により、厚生労働省は新型コロナウイルスに関するセルフチェックには「研究用(=雑品)」でなく「医療用(=体外診断用医薬品)を用いるよう啓蒙しているのですね。[5]
なお、体外診断用医薬品で、検体の採取に医療機器(ランセット、剥離子、綿棒(捲綿子/けんめんし)等)を使用する場合がありますが、その場合の多くはセットで医薬品と医療機器を組み合わせた「コンビネーション製品」として承認申請をしているものと思われます。
検査機関を介す検査キット
家庭で採取した検体を検査機関に送付して、後日結果の送付を受けるタイプがこれです。
例えば郵送での検査サービス(新型コロナウイスルのPCR検査サービス含む)がこれにあたります。
検体の採取に医療機器(ランセット、剥離子、綿棒(捲綿子/けんめんし)等)を使用する場合があります。
家庭用検査キット/サービスの留意点
家庭用検査キットに関しては、上述の通り薬機法が一つのポイントとなりますが、もうひとつ重要なポイントがあります。医師法です。
医師法17条にて、「医師でなければ、医業をなしてはならない。」と定められています。
家庭用検査キット/サービスがこの「医業」に抵触するか否かが、ビジネスを行う上では最重要ポイントと言っても過言ではないでしょう。
(なお、明文での記載は確認されませんでしたが、厚生労働省が発出する通知群を見る限り、現在において「診断・治療」は医師にのみ許される業務であるとみなされていると、私は解釈しています。)
おわりに
冒頭でも申し上げたとおり、今後様々な検査キット/サービスが市場に出てくると思っています。
行政側もそれを見込んで、適切な規制の枠組みとすべく話し合いを進めています[6]。
この分野の事業者さん方は、法の規制の中でどのように自社製品/サービスを展開していくのか、しっかりと検討していくことが必要です。
ここを曖昧にしていると、そもそも通関できなかったりします。
弊事務所ではヘルスケア分野でビジネスを行っていこうとする事業者さんをサポートしておりますので、お困りのことありましたら、お気軽にご相談ください。
参考文献
- 「体外診断用医薬品の取扱いに関する質疑応答集について」平成23年9月6日 薬食機発0906第1号. https://www.pmda.go.jp/files/000159238.pdf. (2022−07−27閲覧)
- 「新型コロナウイルス感染症流行下における薬局での医療用抗原定性検査キットの取扱いについて」令和3年9月27日 厚生労働省厚生労働省新型コロナウイルス感染症対策推進本部 厚生労働省医薬・生活衛生局総務課 事務連絡. https://www.mhlw.go.jp/content/000836277.pdf.(2022−07−28閲覧)
- 「一般事業者からの問合せに対応できる医薬品卸売業者等について(ワクチン・検査パッケージ制度)」.厚生労働省.(更新日不明).https://www.mhlw.go.jp/stf/seisakunitsuite/bunya/0000121431_00296.html. (2022−08−02閲覧)
- 「体外診断用医薬品の一般用検査薬への転用について」平成26年12月25日 薬食発1225第1号. https://www.pmda.go.jp/files/000203308.pdf.(2022−07−28閲覧)
- 「新型コロナウイルス感染症の研究用抗原定性検査キットの販売に関する留意事項について」令和4年5月2日厚生労働省新型コロナウイルス感染症対策推進本部 厚生労働省医薬・生活衛生局監視指導・麻薬対策課 事務連絡. https://www.mhlw.go.jp/content/000935681.pdf(2022−07−28閲覧)
- ”2022年4月11日 薬事・食品衛生審議会 医療機器・体外診断薬部会 議事録".(更新日不明). https://www.mhlw.go.jp/stf/newpage_27045.html.(2022−07−28閲覧)
お気軽にご相談ください。
- 初回相談は無料です。
- 行政書士には秘密保持の義務が課せられております。
- フォームに入力されたメールアドレス以外に、当事務所から連絡差し上げることはいたしません。