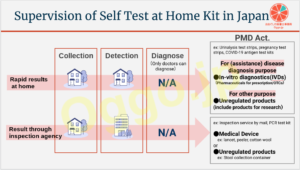
化粧品の「効能の範囲」の変遷を見てみよう!
生活に密着する化粧品の効能の範囲は、薬機法(以前は薬事法という名称でした)とともに、変化を遂げています。
今回はそれを見てみようと思います。
1.薬事法の制定[1][2]
- 1873年(明治06年):「医制」公布、「薬剤取調ノ方法」
- 1943年(昭和18年):戦時中、複数にバラけていた法律をまとめ「薬事法(昭和18年3月12日法律第48号)」を制定
- 1948年(昭和23年):戦後、1943年薬事法を全面改訂した「薬事法(昭和23年7月29日法律第197号)」が制定
- 1960年(昭和35年):1948年薬事法を全面改訂した「薬事法(昭和35年法律第145号)」が制定
この1960年薬事法が、現在の薬機法に継承されていると言われています。
ですので、ここから化粧品の効能の範囲の変遷を見ていこうと思います。
2.化粧品の効能の範囲(1960年/昭和35年)
このときの化粧品の効能の範囲は通知[3]の別表第1にて示されています。
一部だけキャプチャーした画像を貼り付けます。
全体は最下部の「参考文献」の[3]のURLから通知自体をご参照下さい。
類別ごとに分けられており、現在の医薬部外品の効能効果の範囲っぽい表ですね。

3.化粧品の効能の範囲(2000年/平成12年)
平成12年は、化粧品にとって激動の年でした。
大規模な規制緩和によりそれまでの許可制度※を化粧品基準+届出制に変更したことで、新規参入や新製品投入がものすごく簡易になり、化粧品戦国時代の幕開けです。企業責任は大きくなったのですけどね[4][5]。
※昭和61年から2000年までのあいだに、許可制度は次のような変遷を果たしていました。
個別許可制度(個別品目の製造許可)→種別許可制度(化粧品種別許可基準に合致すれば届出で済む)→種別承認制度(種別許可基準外の成分は承認範囲内であれば届出で済む)
このとき改定された化粧品の効能の範囲は、通知[6]の別紙にて示されています。
こちらも一部キャプチャー画像を。
この改正で類別ごとの効能の範囲の設定が廃止され、「化粧品に該当する効能全体を規定する仕組みに改め」られています[6]。もうほとんど、現行のものと変わりませんね。

4.化粧品の効能の範囲(2011年/平成23年〜現在)
このときの化粧品の効能の範囲は通知[7]の別紙にて示されています。
が、これは2000年/平成12年版に、「(56) 乾燥による小ジワを目立たなくする。」が加わっただけですし、検索すればすぐ出てくる情報なので、キャプチャーは割愛します。
さいごに
上記のように化粧品の効能の範囲は変遷を遂げています。
注意しなくてはならないのは、「効能の範囲」であり、「広告の範囲」ではないところ。
ここを履き違えて商品展開してしまうと、景品表示法への抵触や、消費者に使用原料や使用技術へ誤解・ネガティブイメージを与えたりと、長期的ダメージに繋がることも。
化粧品の広告についてはまた別のトピックでお伝えしようと思います。
弊事務所では化粧品事業のサポートを積極的に行っております。
幅広いサポート提供が自慢ですので、お悩みのことがありましたらお気軽にご相談下さい。
初回のご相談は無料で承っております。
参考文献
- 宮本法子「薬事衛生の歴史的変遷と薬学教育6年制改革の開始」日本医史学雑誌第54巻第3号. 305-306. 305. http://jsmh.umin.jp/journal/54-3/305.pdf. (参照2022−08−04)
- 城下賢一「薬事法改正と行政・団体関係1956-1960 : 自民党政権初期の政策決定過程の事例分析」. 立命館大学人文科学研究所紀要 124号. 169-203. 169,170. https://ritsumei.repo.nii.ac.jp/?action=pages_view_main&active_action=repository_view_main_item_detail&item_id=14219&item_no=1&page_id=13&block_id=21. . (参照2022−08−04)
- 昭和36年2月8日付け薬発第44号薬務局長通知「薬事法の施行について」https://www.mhlw.go.jp/web/t_doc?dataId=00ta6817&dataType=1&pageNo=1. (参照2022−08−04)
- 岩倉 良平「薬事法と規制緩和」色材協会誌74 巻 (2001) 5 号. 247-257. https://www.jstage.jst.go.jp/article/shikizai1937/74/5/74_247/_pdf/-char/ja(参照2022−08−04)
- 平成12年9月29日付け医薬発第990号厚生省医薬安全局長通知「化粧品規制緩和に係る薬事法施行規則の一部改正等について」https://www.mhlw.go.jp/web/t_doc?dataId=00ta6887&dataType=1&pageNo=1. (参照2022−08−04)
- 平成12年12月28日付け医薬発第1339号医薬安全局長通知「化粧品の効能の範囲の改正について」https://www.city.hirakata.osaka.jp/cmsfiles/contents/0000002/2816/66845.pdf. (参照2022−08−04)
- 平成23年7月21日付け薬食発0721第1号厚生労働省医薬食品局長通知「化粧品の効能の範囲の改正について」https://www.mhlw.go.jp/web/t_doc?dataId=00tb7518&dataType=1&pageNo=1. (参照2022−08−04)
お気軽にご相談ください。
- 初回相談は無料です。
- 行政書士には秘密保持の義務が課せられております。
- フォームに入力されたメールアドレス以外に、当事務所から連絡差し上げることはいたしません。