消毒用エタノールにかかる規制
手指の消毒に使用する製品は、新型コロナウイルスの感染拡大に合わせて爆発的にニーズが高まりました。
現在では供給が追いつき、入手も容易となってイチ消費者としてもほっとできる状態です。
しかし、インターネットで検索してみると、多くのリサーチ会社は、今後も消毒市場は成長を続けることを予測しているようです(Google.co.jpにて、「消毒 市場 2022」を検索)。
今回は、「手指の消毒に使用する製品のうち、エタノールを主剤とした製品」にスポットを当て、規制周りを見てみたいと思います。
エタノール製品にかかる規制
エタノール製品にかかる規制は多重構造になっています。ざっくりまとめてみました(参考2及び3)。
実際はこの図を頭においておいて、取り扱う製品ごとに規制の範囲か否かを個別確認していきます。
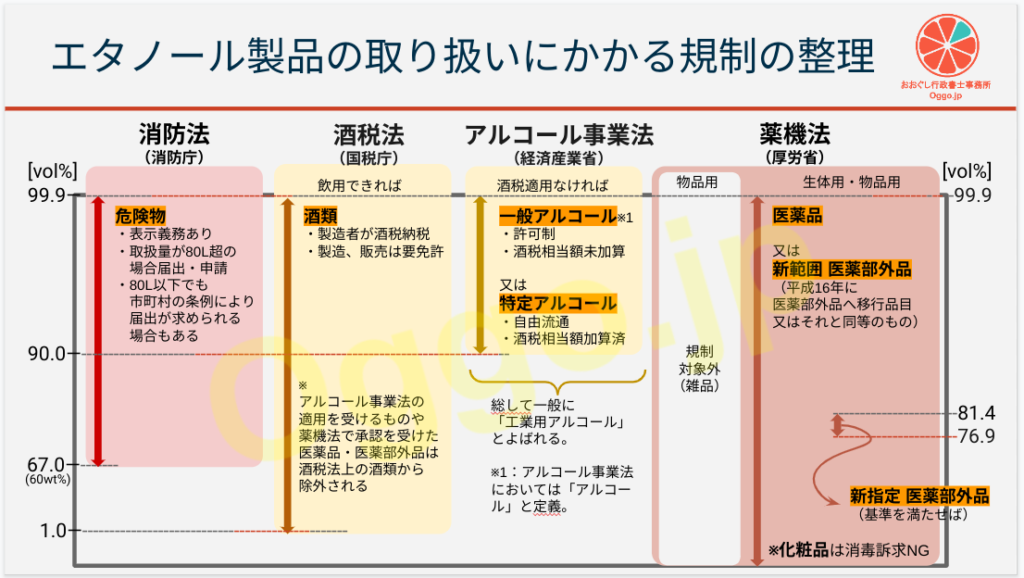
- 消防法(消防庁)における危険物
- 酒税法(国税庁)における酒類
- アルコール事業法(経済産業省)における一般アルコール又は特定アルコール
- 薬機法(厚労省)における医薬品又は医薬部外品(新範囲/新指定)あるいは規制対象外の雑品
人体の消毒目的の場合はどの選択肢を選ぶか→新指定医薬部外品が手堅い
人体に使用する目的の消毒剤は、医薬品又は医薬部外品として薬機法の規制下となります。
化粧品では殺菌、消毒は訴求できません。
考え方としては、「基本的には医薬品だが、医薬部外品の枠内に入れば医薬部外品」です。
単純に比較できるものではありませんが、あえて言うならば難易度は医薬品>>>>>医薬部外品です。
ここでは医薬部外品として、消毒用エタノールが入りうる枠として以下の2つを紹介します。
- 新範囲医薬部外品(厚生労働大臣が指定する医薬部外品のうち、「(9)殺菌消毒薬」)
- 新指定医薬部外品(厚生労働大臣が指定する医薬部外品のうち、「(15)すり傷、切り傷、さし傷、かき傷、靴ずれ、創傷面等の消毒又は保護に使用されることが目的とされている物」)
1.新範囲医薬部外品
新範囲医薬部外品の枠は、通知にて示されています。(「新範囲医薬部外品の製造販売承認申請等に係る取扱いについて」平成20年11月14日付け薬食審査発第1114001号)
簡単に言うと、「有効成分の組合せ及び分量」が既承認の新範囲医薬部外品の範囲内で、かつ既承認の新範囲医薬部外品と「区分(消毒用エタノールが該当するのは「殺菌消毒薬」)「効能又は効果」「用法及び用量」「剤型等」が同一であることが求められます。
基本的に、新範囲医薬部外品の既承認情報を持っていないと、このカテゴリでの申請は難しいと言えるでしょう。
2.新指定医薬部外品
新指定医薬部外品の枠は、「外皮消毒剤 製造(輸入)承認基準」として示されています。
消毒用エタノールの場合、エタノールを76.9〜81.4vol%の範囲で配合すること等が求められます。
難易度は低いとは言えないものの、「人体に使う消毒用アルコールを作って売る」となったときは、やはりこの選択肢が順当かなと思います。
物品の消毒・殺菌目的にも、医薬部外品の枠ができました
従来、人体の消毒に使用するものは医薬部外品、物品は雑品(ただし医療用の器具の殺菌は別)という大まかな区分けがありましたが、
令和4年10月4日に課長通知にて、物品の消毒・殺菌を目的とした製品(下記)も医薬部外品として取り扱う臨時的・特例的な対応が示されました。[5]
- 家具・器具・物品等の消毒・殺菌
- 哺乳びん・乳首の消毒・殺菌
- 調理器具、食器の消毒・殺菌
- 室内の消毒・殺菌
- 浴室・便所の消毒・殺菌
臨時的とされていますが、同時に、今後この取扱について告示が行われる予定であることも示されており、一時的な対応では無いだろうことが予想されます。
(※本項は、2023年2月7日に追記されました)
補足)高濃度エタノール製品について
現在「高濃度エタノール製品」と呼ばれるものが存在することをご存じの方もおいでかもしれません。
これは、「高濃度エタノールを含有するため消毒効果が期待できる」ものの「薬機法上の承認を得ていない」ものです。
コロナ禍での需要逼迫な状況を改善するため薬機法の臨時・特例対応として「やむを得ない場合に限り、医療機関等において高濃度エタノール製品を消毒用アルコールの代替として使用可」(参考4)とされたものですので、需要逼迫の状況が改善された後、撤回される可能性は否定できません。
よって、これからこの枠内でのビジネスを始めることは、長期的に見ておすすめ致しません。
おわりに
弊事務所は感染症の拡大防止に取り組んでいらっしゃる事業者の皆さまを応援しています。
医薬部外品にかかる一連の手続きについてはもちろん、医薬部外品にするしないの判断がつけられない!といったご相談もお受けしております。
最初の面談は無料ですので、企画段階からでも、手元に製品出来上がっていても、どうぞお気軽にご連絡ください。
参考
- "PMDAにおける新型コロナウイルス感染症対策に係る活動について". 独立行政法人医薬品医療機器総合機構. 更新日不明. https://www.pmda.go.jp/about-pmda/news-release/0012.html. (参照2022−06−16)
- "Q1 酒類の定義を教えてください。". 国税庁.更新日不明. https://www.nta.go.jp/taxes/sake/qa/01/01.htm. (参照2022−06−17)
- "アルコール事業法の理解を深める". 経済産業省製造産業局アルコール室. 平成26年3月. https://www.meti.go.jp/policy/alcohol/alc_pamphlet_rev.pdf. (参照2022−06−17)
- 「新型コロナウイルス感染症の発生に伴う高濃度エタノール製品の使用について」令和2年3月23日厚生労働省医政局経済課 厚生労働省医薬・生活衛生局医薬品審査管理課 厚生労働省医薬・生活衛生局監視指導・麻薬対策課 事務連絡
- 令和4年10月4日薬生薬審発1004第2号「物品の消毒・殺菌を目的とする消毒剤たる医薬部外品の製造販売承認申請の取扱いについて」
※当ページは2023年2月7日に、画像の差し替え、「物品の消毒・殺菌目的にも、医薬部外品の枠ができました」項の追加を行いました。
お気軽にご相談ください。
- 初回相談は無料です。
- 行政書士には秘密保持の義務が課せられております。
- フォームに入力されたメールアドレス以外に、当事務所から連絡差し上げることはいたしません。

“消毒用エタノールにかかる規制” に対して2件のコメントがあります。
コメントは受け付けていません。